Ana Y. Estevez
Characterized by a gradual disintegration of the neuronal network, neurodegeneration lies at the crossroads of numerous pathologies such as Parkinson’s, Alzheimer’s disorders, multiple sclerosis, stroke, and others. While the cause of neurodegeneration is often multifactorial in nature (e.g., genetic, age, environmental factors), oxidative damage tends to be a common denominator in the process. The damage is frequently caused by reactive oxygen species (ROS) and free radicals – molecules, containing unstable atoms of oxygen with unpaired electrons that seek to stabilize themselves at the expense of stealing electrons from nearby molecules (e.g., lipids, proteins, nucleic acids). This process is often referred to as oxidative stress. Cellular stressors accompany everyday lives, and humans developed natural mechanisms of combating stress at molecular and cellular levels. The issue, however, arises when endogenous antioxidant systems are compromised, and potent oxidants such as reactive oxygen species (ROS) prevail in nerve cells, leading to cell damage. Thus, understanding the mechanisms of oxidative stress and finding treatment is an overarching goal that would benefit many neurodegenerative disorders.
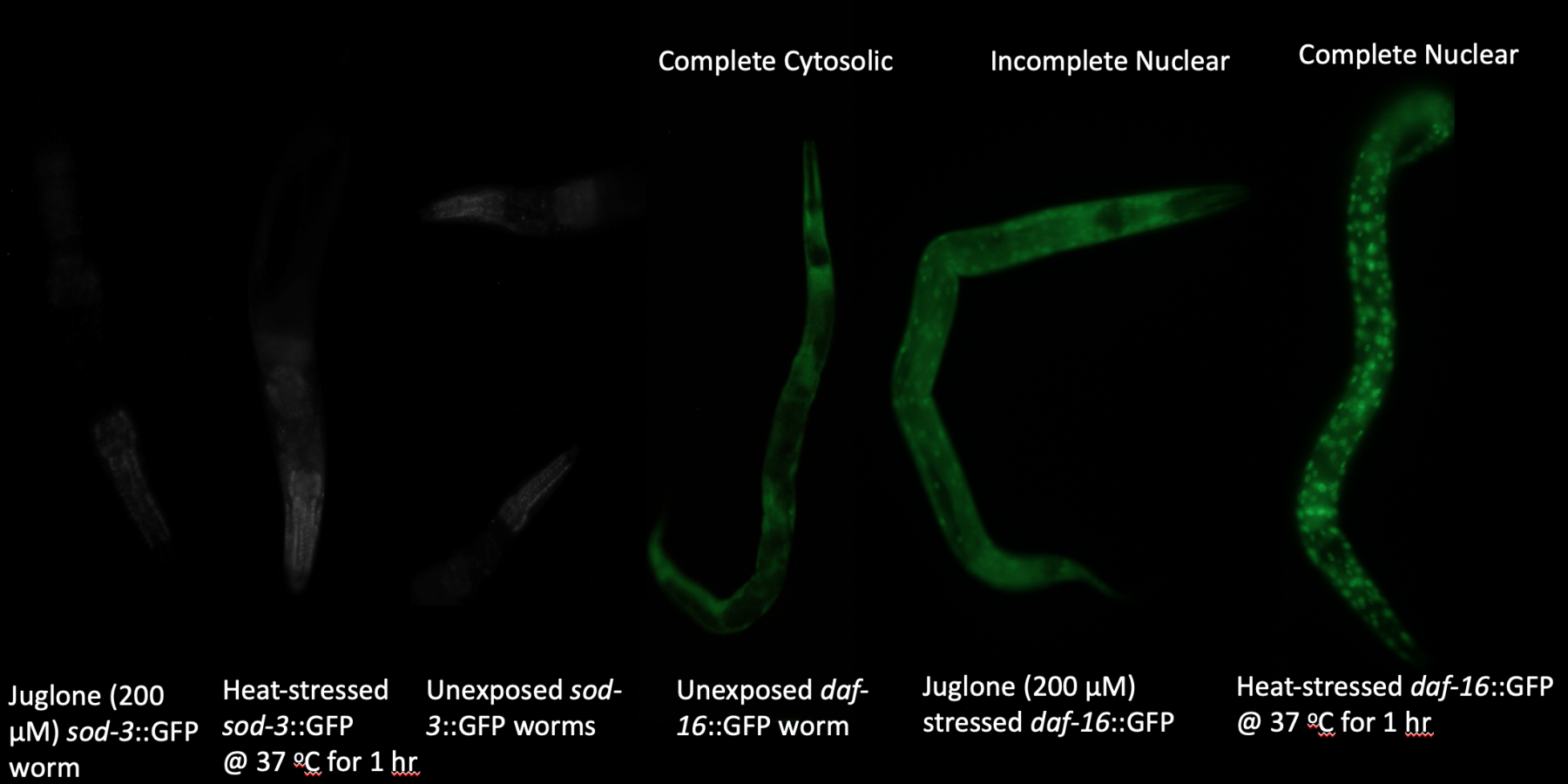
In recent years, advances in nanotechnology and nanotherapeutics provided unparalleled advantages for drug delivery and retention. One example of nanotechnology is nanoparticles defined as tiny particles of a compound like metal oxides with sizes ranging between 1 and 100 nanometers (nm). At Dr. Estevez's lab, we aim to test the therapeutic or toxic effects of novel custom-designed cerium oxide nanoparticles (nanoceria). In theory, cerium oxide nanoparticles of 2 nm in size, allowing them to cross blood-brain barrier, can scavenge ROS, thereby mimicking the activity of endogenous antioxidants. We employ a nematode roundworm, C. elegans, as a model that has emerged as a versatile organism for research in neurobiology due to its inexpensive maintenance, quick lifespan, easily inducible oxidative stress, and availability of transgenic strains. The experimental design of the current study involves the use of two fluorescence markers of oxidative stress such that oxidative stress induction would cause a punctate pattern of fluorescence (qualitative marker) and an increase in fluorescence intensity (quantitative marker). We hypothesized that if nanoceria has therapeutic properties, we would observe a pattern and intensity of fluorescence in stressed worms being similar to unstressed animals, as nanoparticle treatment would help the elimination of ROS with reduced activation of endogenous antioxidants. By monitoring the pattern and intensity of fluorescence in nanoceria-fed worms in vivo after exposure to the oxidative stress-inducing chemical, we can test the potency of different concentrations of nanoparticles to scavenge ROS. In this way, nanoceria supplements can potentially foster neutralization of accumulated ROS in numerous pathologies to reduce collateral damage to the nervous system.